Both, Bruker guidelines and the User Requirements Specification (URS) of DSI-pharm have been fulfilled to reach GMP compliance.
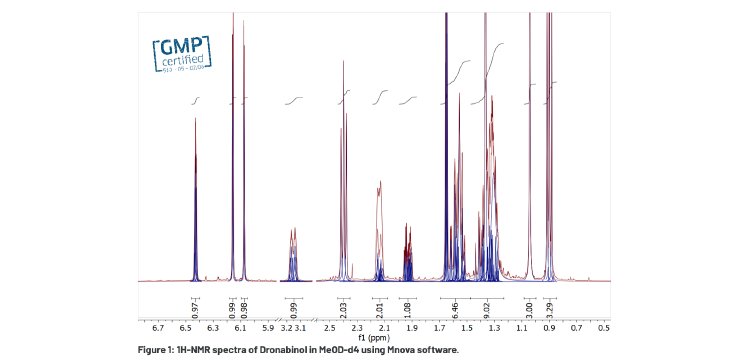
Using NMR as an analytical technique offers a rapid, convenient and cost effective method applicable to all steps of industrial processes (Table 2). Right from the beginning of the development phase through the test phase to the commercial production, all analytical methods in the pharmaceutical and biopharmaceutical industries must be carefully designed, appropriately validated and operated by trained and competent personnel. The dedicated NMR-Team of DSI-pharm knows how to help clients in analytical questions through every step of the pharmaceutical product development and in questions related to product controlling. DSI-pharm already proved its expertise e.g. in structure elucidation, purity determination (according to Ph. Eur. 2.2.33) or long term stability testing (Table 1).
Get in touch with our experts and benefit from this efficient technology!