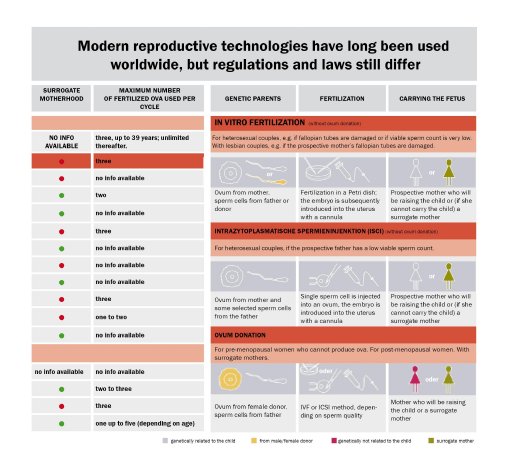
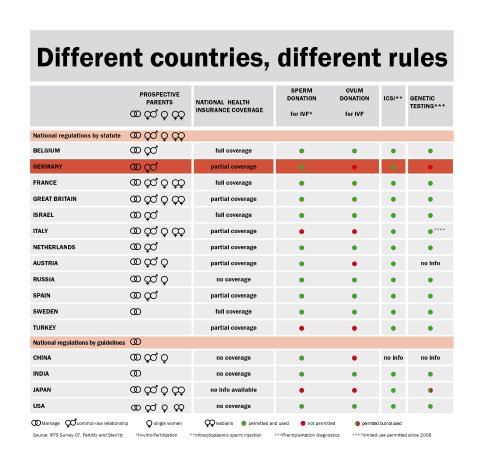
In vitro fertilization (IVF) is often the only hope for couples to fulfill their greatest wish after many years of nerve-racking disappointments. The world's first successful "test-tube" baby was born on July 25, 1978, and the IVF method has now become a standard procedure. Currently, about one in 80 children in Germany are conceived outside the mother's womb by combining the ova and sperm in vitro and then implanting the fertilized egg in the uterus. This method is by no means uncontroversial, however. In Germany, the 'Embryonenschutzgesetz' (Embryo Protection Act) clearly defines the legal parameters. In contrast to the practice in other countries, where all fertilized egg cells are allowed to develop into embryos, Germany does not permit the creation of more than three embryos for the transfer. The selection must be made from among the fertilized egg cells, and the resulting embryos must be implanted. The following figures demonstrate that this clearly reduces the chances for success; although the IVF success rate exceeds 30% worldwide, it is only 26% in Germany. This makes it even more important to improve the technical conditions for success. And it is here that the quality of the incubator is a decisive factor. The incubator performs the function of the mother's womb, perfectly replicating the required conditions in terms of the CO2 atmosphere, temperature and humidity in order to create optimum growth conditions for the cells. At the same time, contamination must be totally avoided so to prevent damaging the highly sensitive cells.
Another benefit of the BINDER incubator is its size, a compact 53-liter configuration that saves precious laboratory space. Its efficient design and small volume produce an optimal space/benefit ratio, making it the preferred equipment for IVF and cell cultivation. Compared to larger chambers that can simultaneously store a multitude of samples, this smaller format creates the type of undisturbed environment that is ideal for cell growth. The fewer the samples stored in the chamber, the lower the frequency of door openings, and thus sensitive cells can grow in a stable and undisturbed environment. For safety reasons, there is a trend towards using a single incubator for each cell type and person. Although this is naturally more expensive, it prevents cross-contamination and mistakes that can result from sample mix-ups. The inner chamber has integrated, smooth-edged shelf supports, which are formed as part of the inner chamber wall in the deep-draw process, i.e. no sharp corners or edges. The advanced design permits operation without any interior fittings such as fans or filters. This facilitates cleaning and prevents contamination deposits from forming in inaccessible corners. At the same time, the Permadry®-System ensures dry walls, free of condensation, while operating at a relative humidity of more than 95%. The water level can be visually inspected and water replacement is easy. All of these features ensure that the specimens being tested have the best conditions for healthy growth.
The hot air sterilization cycle runs for 10 hours at 180 °C, practically overnight at the push of a button - a highly desirable and time-saving feature for laboratories. BINDER provides additional information about this process on a special website at www.true-sterilization.com . This site also contains useful links to global sterilization standards. This smaller CB model can also compete with the larger 150 and 210 L models in terms of its standard technical features. The CO2/air mixture is injected into the inner chamber through a jet; since the interior is under a slight vacuum, the resulting venturi effect distributes the mixture homogenously. This eliminates the need for a fan, which creates turbulence and complicates cleaning. An infrared sensor selectively measures the CO2 concentration in real-time. A microprocessor controls temperature and CO2 concentration. The standard model includes an automatic self-diagnostic system with optical and acoustic alarm functions as well as a potential-free switchover contact for continuous central monitoring of the CO2 concentration. In the event of any deviations from the setpoints, the system automatically assumes control of the injection process. In addition to IVF, other important areas of application for this new BINDER chamber include cell biology, bio-tissue engineering, human and veterinary medicine, microbiology, dental technology, and the pharmaceutical industry, i.e. all applications with demanding requirements for long-term stability, reliability, homogeneity, and reproducibility of results.